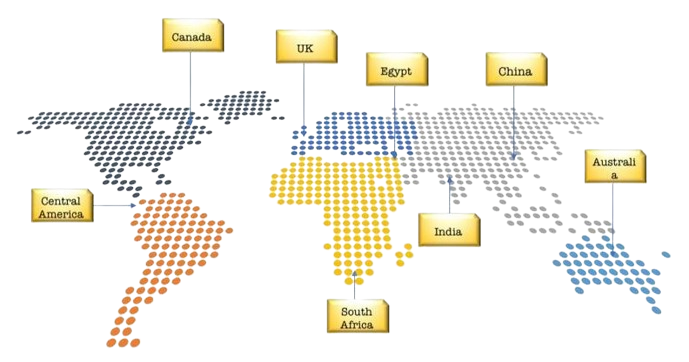
GLOBAL OPERATIONS
Global coverage across every continent – ensuring product reach , market access and regulatory support.
Phone
+44 7448033565
+44 7340667841
Assuring You Our Best Services Through
Kunz Therapeutics Ltd.,
WA145HN, Cheshire,
United Kingdom.
MARKETING SERVICES
We also conduct Clinical meetings on regular basis, enabling us to provide a platform to interact with Physicians, Pharmacist & other Faculty members to understand their views about products. Following services are integral part of our commitment to our clients around the globe.
i. Market Research
Near to accurate market information helps in assessing market potential for specific products. Before we propose any product to importing company, we ensure that we provide him with possible product potential.
ii. Marketing Strategy
Based on market research and inputs from various local sources, we provide suitable and effective marketing strategy. This is part of our brand flagship activity.
iii. Promotional Support
Based on market strategy, we do provide substantial technical data from reliable sources about our products. Whenever possible, we also provide Visual Aid, Product Literatures, Compliments etc.
iv. Marketing Personnel Training
Before product launch, we ensure that all the marketing personnel undergo adequate product training. We also ensure that entire team has common understanding of marketing strategy.
v. Product Launch
Depending upon market and products, official product launch Programs are undertaken.
vi. Clinical Meetings
We also conduct Clinical meetings on regular basis, enabling us to provide a platform to physicians airing their view about products and questions if any.
vii. Post Launch Sales Monitoring
This is an important aspect of our efforts to ensure that products are sold to consumers. This exercise gives us an opportunity to keep us up to date with market requirements and also helps us in addressing product related issues if any.
REGULATORY SERVICES
This is key area of our operations. Without this, we may not be able to move at faster pace. Hence, most of the time, we first prepare country specific registration documents before identifying suitable partner.
I. Registration dossier
Our full fledged regulatory department is always ready to provide you with needed support on this front.
Ii. Country Specific Registration Sample
Dossiers are submitted along with registration samples. Samples are made as per stipulated guidelines of importing country.
iii. Post Submission Follow up
Time is the essence of registration activity. Hence, we do make regular follow-up with regulatory authorities of different countries. Our efforts are in addition to the efforts put in by our client.